Fluoridation
Definition
Fluoridation is the addition of fluoride to water supplies to help prevent tooth decay .
Description
The element fluorine is the seventeenth most abundant element in the earth's crust. It occurs as fluoride ion in combination with other elements such as sodium. Most water supplies naturally contain low levels of fluoride. In much of the United States, as well as in other parts of the world, fluoride is added to community water systems to bring fluoride levels up to the recommended amount for preventing teeth decay: 0.7–1.2 parts of fluoride to 1 million parts of water (parts per million or ppm). The levels of naturally occurring fluoride in fresh water range from less than 0.1 ppm to more than 13 ppm. Seawater contains about 1.5 ppm. As of 2000, about 162 million Americans—two-thirds of the population—were served by fluoridated water systems.
Mode of action
Systemic fluorides, including fluoridated water and prescription fluoride supplements supplied as tablets, drops, or lozenges, can be incorporated into the enamel of children's developing teeth. The enamel that covers the crown, the part of the tooth that is above the gum, is made of a substance called hydroxyapatite. When enough fluoride from water, supplements, food, or other sources enters the bloodstream and reaches the teeth while the enamel is forming, the fluoride can replace a piece of the hydroxyapatite molecule to form fluorapatite. Thus fluoride becomes part of the tooth enamel. Fluoride makes the tooth more resistant to acids produced by the bacteria that cause tooth decay. These acids dissolve the enamel, causing cavities. Fluoride in the enamel appears to do the following:
- make the enamel less susceptible to bacterial acids
- reduce the bacteria's ability to produce acid
- reduce the number of bacteria in plaque deposits
It is unlikely that sufficient fluoride will be incorporated into the enamel throughout the years of crown formation. With optimally fluoridated water (1 ppm) as the primary source of fluoride, a child would have to drink two quarts of water every day for 12 to 14 years to incorporate fluoride into all of the baby and adult teeth as they form. The child would be ingesting about 2 mg. of fluoride daily.
Topical fluorides are applied directly to the surfaces of fully-formed teeth. Fluoridated water acts as a topical—as well as a systemic—source of fluoride. Other topical fluorides include fluoridated toothpastes and mouthwashes and fluoride gels that are applied to children's teeth at dental examinations. Topical fluoride is the most effective mineral for renewing or remineralizing the surface layers of enamel and dentin as they wear out and are eaten away by acids from food and bacteria. Fluoride remineralization makes the tooth surface more resistant to decay and reverses early decay processes. Thus topical fluorides help prevent decay in both children and adults. Systemic fluorides also can provide topical protection because they are incorporated into the saliva that bathes the teeth.
Sources of fluoride
Fluoridated water is a major source of fluoride. Most bottled water contains only trace amounts of fluoride. Filtered water and well water vary greatly in their fluoride content. Children who drink water that is low in fluoride may be given fluoride supplements.
Fluoride occurs in many different foods and is also added to some foods. Fruits and vegetables may contain more than 0.2 mg of fluoride per serving, depending on where they were grown and whether fluoridated water was used for irrigation and processing. Most seafood is high in fluoride. The amount of fluoride in beverages depends on the amount of fluoride in the water used to make them. Many vitamins and medicines also contain fluoride. Most baby food is made with nonfluoridated water.
Fluoridated toothpastes and mouthwashes contain high amounts of fluoride. A tube of fluoridated toothpaste may contain as much as 1 to 2 gm of fluoride. Nonprescription mouthwashes can contain up to 120 mg of fluoride. Children between the ages of two and six swallow about 33 percent of the toothpaste they use; children between seven and 15 swallow about 20 percent. The average child using the typical amount of fluoridated toothpaste will swallow or absorb 0.5 to 1.0 mg. of fluoride per brushing. Much of this fluoride is excreted.
It is believed that fluoridated water is between 20 and 60 percent effective in preventing cavities in children and adults. Early studies suggested that water fluoridation was eliminating tooth decay in children. However, other factors are recognized in the early 2000s as having contributed to the decline in dental cavities. The widespread use of fluoridated toothpastes and mouthwashes has increased children's sources of fluoride significantly. Furthermore, both children's and adults' knowledge about dental care and dental hygiene has improved in the last quarter of the twentieth century.
General use
History
In the early twentieth century a young dentist in Colorado Springs, Colorado, named Frederick McKay, noticed that many local residents had brown stains on their permanent teeth and that their teeth were surprisingly resistant to decay. McKay eventually discovered that this "mottling"—as he called it—resulted from high levels of naturally occurring fluoride in the drinking water.
The first fluoridation of a public water system took place in Grand Rapids, Michigan, in 1945. By the 1950s and 1960s increasing numbers of communities were fluoridating their water using by-products from the phosphate fertilizer industry. The practice became mired in controversy, and it remains so in the first decade of the twenty-first century. Since the decision to fluoridate usually is made at the local level, by public officials or a vote of the people, fluoridation has become a political as well as a scientific controversy.
Proponents
Proponents of water fluoridation argue the following:
- It significantly reduces tooth decay, both before and after tooth enamel has formed.
- The fluoride levels used are completely safe.
- The children of parents who are poorly informed about dental hygiene and cavity prevention or who cannot afford dental treatment are still protected against tooth decay.
Most government agencies and scientific and professional organizations agree that water fluoridation is safe and effective in preventing tooth decay and cost-effective in that it reduces the need for expensive dental treatment. Among the organizations that endorse fluoridation of water supplies are the following:
- American Academy of Pediatric Dentistry
- American Dental Association
- American Medical Association
- National Institute of Dental and Craniofacial Research
- U.S. Centers for Disease Control and Prevention (CDC)
- U.S. Public Health Service
- World Health Organization (WHO)
Opponents
Opponents of water fluoridation often use one or more of the following arguments:
- Any fluoride above the naturally occurring (usually trace) amounts is unnecessary and possibly toxic.
- An individual dose of fluoride cannot be controlled because it depends on the amount of fluoridated water that a child ingests each day.
- Fluoridation of public water systems deprives people of freedom-of-choice as to what they ingest.
- People can choose from a variety of fluoride-containing products that are just as effective as fluoridated water.
- Where the water is not fluoridated, schools often provide fluoridation programs, and parents can choose whether their children participate.
- Although fluoride may help prevent decay, good diet, good oral hygiene , and regular dental cleanings can be just as effective.
- Fluoride can be toxic and even fatal at higher doses.
- The difference between the amount of fluoride that is beneficial and the amount that can cause mottling is only two to four-fold.
- People vary in their susceptibility to the effects of fluoride.
- It is impossible to determine how much fluoride a child is ingesting because of the numerous sources of fluoride in food and products; a child may regularly drink water from sources with different fluoride levels.
- Fluoride is ineffective against gum disease, the major destroyer of teeth.
Communities throughout the United States, as well as many countries, have chosen not to fluoridate their water. In the early 2000s a number of countries have discontinued fluoridation because of ongoing concerns about possible health effects.
Fluoride dosages
There is some disagreement as to whether fluoride is an essential mineral in humans. Relatively low levels of fluoride (20–80 mg) are considered toxic. Less than 1 gm of fluoride can be fatal to a small child. The Food and Nutrition Board of the Institute of Medicine of the U.S. National Institutes of Health has determined an adequate daily intake of fluoride and a maximal safe daily intake, based on a child's weight:
- infants up to six months of age or about 16 lb (7 kg): 0.01 mg is adequate and 0.7 mg is the maximum safe intake
- infants between six and 12 months or about 20 lb (9 kg): 0.5 mg and 0.9 mg
- children one to three years of age or about 29 lb (13 kg): 0.7 mg and 1.3 mg
- children aged four to seven or about 48 lb (22 kg): 1.0 mg and 2.0 mg
- children aged nine to 13 or about 88 lb (40 kg): 2.0 mg and 10 mg
- children aged 14 to 19 or about 125–166 lb (57–76 kg): 3.0 mg and 10 mg
Fluoride supplements often are prescribed for children who drink nonfluorinated water and do not use fluoride toothpaste. Fluoride supplements should not be used if the drinking water contains more than 0.6 ppm of fluoride. One ppm of fluoride is equivalent to about 1 mg per quart (or liter) of water. Fluoride supplements should not be given to babies under six months of age regardless of the fluoride content of the water. Babies get adequate fluoride from breast milk or infant formula. Powdered or concentrated infant formula should be mixed with low-fluoride or fluoride-free water.
If the water supply contains 0.0 to 0.3 ppm fluoride, the recommended daily dosage of fluoride supplement is:
- 0.25 mg for children aged six months to three years
- 0.50 mg for children aged three to six years
- 1 mg for children aged six to 16 years
If the water supply contains 0.3 to 0.6 ppm fluoride, the recommended daily dosage of fluoride supplement is:
- 0.0 mg for babies aged six months to three years
- 0.25 mg for children aged three to six years
- 0.50 mg for children aged six to 16
Fluoride supplements usually come in the form of sodium fluoride: 2.2 mg of sodium fluoride supplies 1 mg of fluoride ion.
Precautions
A child easily can swallow enough fluoridated toothpaste to exceed the recommended daily amount of fluoride by four-fold. A medium-sized toothpaste tube contains enough fluoride to make a child seriously ill or even cause death should the child eat it all. The flavorings added to toothpaste to encourage children to brush also can entice them into eating it. Toothpaste always should be stored out of the reach of children.
Side effects
As little as four to eight mg of fluoride ingested daily while the tooth enamel is forming can cause mottling—often called fluorosis—in children under age eight. Fluorosis only affects children whose teeth are still developing within the gums. Symptoms of fluorosis include:
- teeth discoloration
- white or brown chalky spots
- brown enamel
- pitting of teeth
- excessive wear on the enamel
- structural damage to the enamel
- brittle teeth in which the enamel breaks easily
The extent of mottling depends on the following:
- when the excess fluoride is ingested
- how much is ingested
- over how long of a period it is ingested
- how much of the fluoride reaches the enamel
Most cases of fluorosis are very mild. Very mild to mild fluorosis has no effect on tooth function and may help prevent decay. Even severe fluorosis is not harmful. Fluorosis in children appears as of 2004 to be increasing; however, it is not known whether this is from water fluoridation, the excessive use of fluoride-containing products, or both.
Prevention
No type of fluoridation can replace good dental care and hygiene, which are necessary for preventing gum disease as well as tooth decay. Weekly rinsing with a fluoride mouthwash can reduce decay in children by 20–40 percent. Fluoride supplements can reduce decay
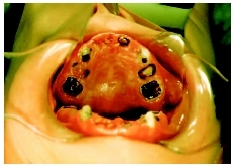
Topical fluoride treatments given in a dentist's office have been proven to be 40 percent or more effective in preventing decay. These treatments include fluoride gels and foams and fluoride varnishes. Advantages of fluoride varnishes include the following:
- They are more concentrated than other fluoride treatments and so are less likely to be ingested.
- They are fast and easy to apply.
- They continue to provide fluoride to the enamel for about 24 hours after application.
Varnishes may be particularly appropriate for young children and those with special needs since varnishes do not require the use of a fluoride tray.
Parental concerns
It is the parents' responsibility to monitor their child's fluoride intake. Pregnant and nursing mothers should pay close attention to how much fluoride they ingest. Children should:
- be over two years of age before using a fluoridated toothpaste; younger children are likely to swallow most of their toothpaste
- use a pea-sized amount of toothpaste or less
- not use fluorinated mouthwashes until the age of six
- be prevented from swallowing fluoridated toothpaste or mouthwash
KEY TERMS
Dentin —The middle layer of a tooth, which makes up most of the tooth's mass.
Enamel —The hard, outermost surface of a tooth.
Fluorapatite —Fluoride-substituted hydroxyapatite.
Fluorosis —Mottled discoloration of tooth enamel due to excessive systemic ingestion of fluoride during tooth development.
Mottling —Fluorosis; spotting on the teeth due to excess fluoride as the tooth enamel is forming.
Plaque —A deposit, usually of fatty material, on the inside wall of a blood vessel. Also refers to a small, round demyelinated area that develops in the brain and spinal cord of an individual with multiple sclerosis.
Remineralization —Recalcification; the process by which minerals from saliva and food are added to the surface of the enamel or to the dentin.
Systemic absorption —Any substance topical, inhaled, or ingested that is absorbed into the blood-stream and distributed throughout the body.
Topical —Not ingested; applied to the outside of the body, for example to the skin, eye, or mouth.
See also Dental development .
Resources
BOOKS
Bailey, K., et al. Fluoride in Drinking Water. London: IWA, 2002.
Bryson, Christopher. The Fluoride Deception. New York: Seven Stories Press, 2004.
PERIODICALS
"Anti-Fluoride Flood." Better Nutrition 65, no. 1 (January 2003): 30–1.
Gorman, Megan Othersen. "Got Enough Fluoride?" Prevention 54, no. 2 (February 2002): 48.
Mascarenhas, Ana Karina. "Risk Factors for Dental Fluorosis: A Review of the Recent Literature." Pediatric Dentistry 22, no. 4 (2000): 269–77.
Motavalli, Jim. "Your Health: Facing up to Fluoride." E: the Environmental Magazine 12, no. 1 (January/February 2001): 40–1.
Pendrys, D. G. "Risk of Enamel Fluorosis in Nonfluoridated and Optimally Fluoridated Populations: Considerations for the Dental Professional." Journal of the American Dental Association 131 (2000): 746–55.
Pratt, Edwin, Jr., et al. "Fluoridation at Fifty: What Have We Learned?" The Journal of Law, Medicine, and Ethics 30, no. 3 (Fall 2002): 117–22.
"Water Fluoridation Debate." Journal of Environmental Health 65, no. 3 (October 2002): 52.
ORGANIZATIONS
American Academy of Pediatric Dentistry. 211 East Chicago Avenue, Suite 700, Chicago, IL 60611–2663. Web site: http://www.aapd.org.
American Dental Association. 211 East Chicago Avenue, Chicago, IL 60611–2678. Web site: http://www.ada.org.
Fluoride Action Network. PO Box 5111, Burlington, VT 05402. Web site: http://www.fluoridealert.org.
National Center for Fluoridation Policy and Research. Department of Oral Biology, School of Dental Medicine, State University of New York at Buffalo, Buffalo, NY 14214–3008. Web site: http://fluoride.oralhealth.org .
WEB SITES
Connett, Paul. "The Absurdities of Water Fluoridation." Redflagsdaily.com , November 28, 2002. Available online at http://www.redflagsweekly.com/connett/2002_nov28.html (accessed December 27, 2004).
"Fluoride." American Academy of Pediatric Dentistry . Available online at http://www.aapd.org/publications/brochures/content/floride.html (accessed December 27, 2004).
"Oral Health Topics: Fluoride & Fluoridation." American Dental Association. Available online at http://www.ada.org/public/topics/fluoride/fluoride_article01.asp (accessed December 27, 2004).
Margaret Alic, PhD
Comment about this article, ask questions, or add new information about this topic: